 
The average age at first hospitalization was 93 days, and at diagnosis, 141 days. (1993) reported 36 patients with T-, B- SCID among 117 patients with SCID. The 3 probands died of pneumonia in the first 3 years of life. (1967) reported 3 families in which multiple sibs had congenital hypogammaglobulinemia that defied classification. (1967) reported that 'thymic alymphoplasia,' now known as thymic dysplasia ( Nezelof, 1992), was frequent among Mennonites living in southern Manitoba. Furthermore, the thymus, which was usually normal in Bruton agammaglobulinemia, was very small with absence of lymphoid cells and Hassall corpuscles. In contrast to X-linked Bruton agammaglobulinemia, patients were unusually susceptible to fungal and viral as well as pyogenic pathogens, lacked delayed hypersensitivity, and showed failure of antibody production. Good (1964) referred to the recessive form of agammaglobulinemia as the Swiss type. The findings were consistent with 'thymic dysplasia' ( Nezelof, 1992). Affected patients had a small thymus with depletion of lymphoid cells, suggesting a failure or arrest in embryologic development of the gland. 
Tobler and Cottier (1958) reported families with agammaglobulinemia and lymphopenia showing autosomal recessive inheritance. Nezelof (1992) noted that 'Swiss-type agammaglobulinemia' is a historic term referring to severe combined immunodeficiency as a disorder with both agammaglobulinemia and T-cell lymphopenia, and does not represent a single disease entity. At that time, the disorder was termed 'Swiss-type agammaglobulinemia' to distinguish it from the less severe Bruton agammaglobulinemia ( 300755) in which T lymphocytes are unaffected. (1968) reported a form of congenital immunodeficiency with agammaglobulinemia and absence or decreased numbers of lymphocytes. Hitzig and Willi (1961), Hitzig (1968), and Hitzig et al. Glanzmann and Riniker (1950) reported 2 pairs of sibs who had severe infections, candidiasis, agammaglobulinemia, and lymphopenia. The most common form of SCID is X-linked T-, B+, NK- SCID (SCIDX1 300400) caused by mutation in the IL2RG gene ( 308380) on chromosome Xq13.1.Īutosomal recessive SCID includes T-, B-, NK+ SCID, caused by mutation in the RAG1 and RAG2 genes on 11p13 T-, B+, NK- SCID ( 600802), caused by mutation in the JAK3 gene ( 600173) on 19p13 T-, B+, NK+ SCID (IMD104 608971), caused by mutation in the IL7R gene ( 146661) on 5p13 T-, B+, NK+ SCID (IMD105 619924), caused by mutation in the CD45 gene (PTPRC 151460) on 1q31-q32 T-, B+, NK+ SCID (IMD19 615617), caused by mutation in the CD3D gene ( 186790) on 11q23 T-, B-, NK- SCID ( 102700) caused by mutation in the ADA ( 608958) gene on 20q13 and T-, B-, NK+ SCID with sensitivity to ionizing radiation ( 602450), caused by mutation in the Artemis gene (DCLRE1C 605988) on 10p13 ( Kalman et al., 2004) and T-, B-, NK+ SCID with microcephaly, growth retardation, and sensitivity to ionizing radiation ( 611291), caused by mutation in the NHEJ1 gene ( 611290) on 2q35.Īpproximately 20 to 30% of all SCID patients are T-, B-, NK+, and approximately half of these patients have mutations in the RAG1 or RAG2 genes ( Schwarz et al., 1996 Fischer et al., 1997).Įarly Descriptions of Autosomal Recessive SCID Presence or absence of NK cells is variable within these groups. 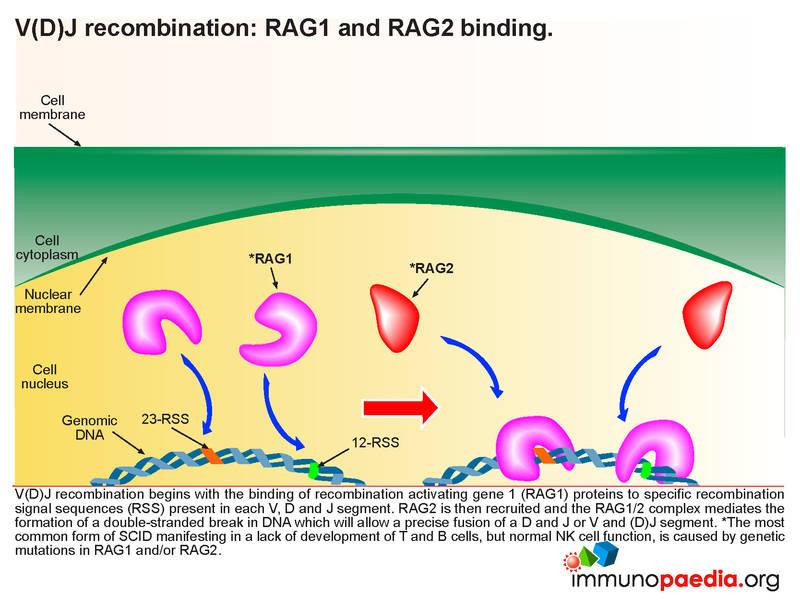
SCID can be divided into 2 main classes: those with B lymphocytes (B+ SCID) and those without (B- SCID). The overall prevalence of all types of SCID is approximately 1 in 75,000 births ( Fischer et al., 1997 Buckley, 2004). Without treatment, patients usually die within the first year of life. The common characteristic of all types of SCID is absence of T cell-mediated cellular immunity due to a defect in T-cell development. Laboratory analysis shows profound lymphopenia with diminished or absent immunoglobulins. Patients with SCID present in infancy with recurrent, persistent infections by opportunistic organisms, including Candida albicans, Pneumocystis carinii, and cytomegalovirus, among many others. Severe combined immunodeficiency refers to a genetically and clinically heterogeneous group of disorders with defective cellular and humoral immune function. 
Severe combined immunodeficiency, X-linked Severe combined immunodeficiency due to ADA deficiency SCID, autosomal recessive, T-negative/B-positive typeīare lymphocyte syndrome, type II, complementation group B Severe combined immunodeficiency, B cell-negativeīare lymphocyte syndrome, type II, complementation group Dīare lymphocyte syndrome, type II, complementation group A Severe combined immunodeficiency, Athabascan type Severe combined immunodeficiency with microcephaly, growth retardation, and sensitivity to ionizing radiation Bare lymphocyte syndrome, type II, complementation group Eīare lymphocyte syndrome, type II, complementation group C 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
